Weathering
As a general rule, each process can be put into one of two chronological categories in terms of when their effect is most significant:
Early stage of a spill: spreading, evaporation, dispersion, emulsification and dissolution
Later stage of a spill: oxidation, sedimentation and biodegradation. These are longer term processes that will determine the ultimate fate of the oil spilled.
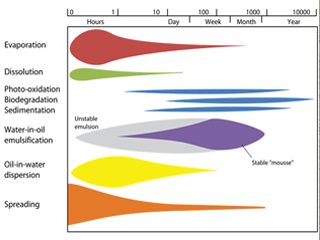
The diagram below represents the fate of a typical crude oil spill, showing changes in the relative importance of weathering process with time (from hours to years). The width of the band indicates the importance of the process.
Spreading

small thickness layer
Spreading over the sea surface begins as soon as oil is spilled.
The speed at which this takes place largely depends upon the viscosity of the oil, which in turn depends both on the oil composition and the ambient temperature. Fluid, low viscosity oils spread more quickly than those with a high viscosity. At low temperature, an oil will tend to be more viscous than at higher temperature as viscosity is inversely proportional to temperature.
Spreading is rarely uniform and large variations in the thickness of floating oil are typical.
After a few hours the slick will typically begin to break up due to the action of winds, wave action and water turbulence, and will often form narrow bands or windrows parallel to the wind direction.
In addition to the oil type, the rate at which oil spreads is determined by prevailing environmental conditions such as temperature, water currents, tidal streams and wind speeds. Higher ambient temperatures, stronger winds and sea surface currents usually result in a more rapid rate of spreading.
Evaporation
The rate of evaporation and the speed at which it occurs depend upon the volatility of the oil.
An oil with a large percentage of light and volatile compounds will evaporate more than one with a larger proportion of heavier compounds. For example, gasoline, kerosene and diesel, which are all light products, tend to evaporate almost completely within a few days. In contrast, little evaporation will occur from a spilled heavy fuel oil.
In temperate climates, those components of the oil with a boiling point under 200ºC tend to evaporate within the first 24 hours. The rate of evaporation can increase as the oil spreads, due to the increased surface area of the slick.
Rougher seas, high wind speeds and high temperatures also tend to increase the rate of evaporation and the proportion of an oil lost to the atmosphere by this process.
Dispersion

can cause the break up of the slick into droplets.
Waves and turbulence at the sea surface can cause some or all of a slick to break up into fragments and droplets of varying sizes. These become mixed into the upper levels of the water column. Some of the smaller droplets will remain suspended in the sea water while the larger ones will tend to rise back to the surface. They may then reform a slick or spread out to form a very thin film.
Dispersed oil droplets have a greater surface area to volume ratio than floating oil. This encourages other natural processes such as dissolution, biodegradation and sedimentation to occur.
The speed at which an oil disperses is largely dependent upon the nature of the oil and the sea state. Dispersion occurs most quickly if the oil is light and of low viscosity and if the sea is very rough (see BRAER case history).
The addition of chemical dispersants can accelerate the process of natural dispersion.
Emulsification

(insert) and close view of emulsified
heavy fuel oil.
An emulsion is formed when two liquids combine, with one ending up suspended in the other.
Emulsification of crude oils refers to the process whereby sea water droplets become suspended in the oil to forma water-in-oil emulsion. This occurs by physical mixing promoted by turbulence at the sea surface. The emulsion formed is usually very viscous and more persistent than the original oil and is sometimes referred to as chocolate mousse because of its appearance. The formation of these water-in-oil emulsions causes the volume of pollutant to increase between 3 and 4 times. This slows and delays other oil weathering processes, and can complicate the response.
Oils with an asphaltene content greater than 0.5% tend to form stable emulsions which may persist for many months after the spill has occurred. Oils containing a lower percentage of asphaltenes are less likely to form water-in-oil emulsions.
Emulsions may separate into oil and water again if heated by the sun under calm conditions or when stranded on shorelines.
Dissolution
Water soluble compounds in an oil may dissolve into the surrounding water. This occurs most quickly when the oil is dispersed in the water column.
Components that are most soluble in sea water are the light aromatic hydrocarbons compounds such as benzene and toluene. However, these compounds are also those first to be lost through evaporation, a process which is 10 -1000 times faster than dissolution. Most crude oils and all fuel oils contain relatively small proportions of these compounds making dissolution one of the less significant processes.
Oxidation

Oils react chemically with oxygen either breaking down into soluble products or forming persistent compounds called tars. This process is promoted by sunlight, but is very slow and even in strong sunlight, thin films of oil break down at no more than 0.1% per day.
The formation of tars is caused by the oxidation of thick layers of high viscosity oils or emulsions. This process forms an outer protective coating of heavy compounds that results in the increased persistence of the oil. Tarballs, which are often found on shorelines and have a solid outer crust surrounding a softer, less weathered interior, are a typical example of this process.
Sedimentation/Sinking

(image courtesy of NOAA)
Very few oils sink in the marine environment.
When floating oil is getting close to the shore, sedimentation can occur. Indeed, once all the lighter compounds have evaporated and the slick has weathered at sea, some oils can be close to the density of seawater. When floating, semi submerged or dispersed oil comes into contact with suspended sediment, the sediment can bind to it. This particularly happens in shallow waters. If the contamination’s heavy, then dense ‘tar-mats’ can form on the seabed near shore.
Oil stranded on sandy shorelines often becomes mixed with sand and other sediments. If this mixture is subsequently washed off the beach back into the sea it may then sink.
In addition, if the oil catches fire or is voluntarily burnt (in-situ burning) after it has been spilled, the residues that sometimes form can be sufficiently dense to sink.
Biodegradation
Sea water contains a wide range of micro-organisms that use hydrocarbons as a source of energy and can partially or completely degrade oil to water soluble compounds and eventually to carbon dioxide and water.
Many types of marine micro-organism exist and each tends to degrade a particular group of compounds in crude oil. However, some compounds in oil are very resistant to attack and may not readily degrade.
The main factors affecting the efficiency of biodegradation, are the levels of nutrients (nitrogen and phosphorus) in the water, the temperature and the level of oxygen present.
As biodegradation of oil requires oxygen, this process can only take place at the oil-water interface since no oxygen is available within the oil itself. The creation of oil droplets through dispersion, increases the surface area to volume ratio of the oil, and therefore increases the area available for micro-organisms to attach to the surface of the oil and for biodegradation to take place.
